GEM$ Award: Grants for Pediatric Emergency Medicine
The GEM$ (Grants for Pediatric Emergency Medicine) Award was inaugurated in 2020 to provide seed funding for high quality research projects for pediatric emergency medicine (PEM) faculty and fellows. The award is intended to spark new scientific achievements of the highest quality and impact, and enhance the research environment at Children's Healthcare of Atlanta. Funds may be used for any pediatric emergency medicine-related research, including basic, clinical, translational, and outcomes research.
Applications are currently being accepted.
All research must be hypothesis driven and describe a discrete project that can be accomplished in the time and budget allotted.
Eligibility
- Faculty and any stage fellow within the Emory Department of Pediatric Emergency Medicine are encouraged to apply.
- Projects must be unfunded and unpublished. Sub-studies of a funded project are eligible as long as the sub-study is not already funded.
Budget
- The period for all projects is Febuary 1 through January 31. Applicants may request a one- or two-year project period.
- Faculty may request a maximum of $5,000 and may not serve as PI on more than one GEM$ award at a time. Smaller budget requests are encouraged.
- Fellows may request a maximum of $2,500 and may only receive one GEM$ award as a fellow. Smaller budget requests are encouraged.
Requirements
- Applicants must propose a hypothesis-driven project and specify an end product of their proposed project, such as a presentation, manuscript, grant application or other scholarly deliverable. For fellows, the end product must be completed by the end of the training program period. All products must be reported in the online grants management system.
- If selected for award, any projects involving human subjects research must have IRB approval prior to receiving an official Notice of Award.
- Proper acknowledgement of this award must be included in all publications, oral and poster presentations, and abstracts that result from this project. Please use the following statement: "This research was supported by Emory Department of Pediatrics, Division of Emergency Medicine and Children’s Healthcare of Atlanta."
- The principal investigator must respond in a timely manner to all requests for project updates, which will be required at least every 6 months.
Selection Criteria
- Applications are reviewed and selected based on the NIH grant application scoring system.
- Please click here to view an explanation of how this system has been adapted for use with the GEM$ Award.
Concept Proposal (PDF): A 3-page, single-spaced, Arial 11 font, 1" margin scientific plan, which includes the following:
- Title of project
- Principal investigator, plus the name(s) of other research team members, as applicable (e.g. mentor, co-investigator, collaborator)
- Background and significance
- Purpose of the research project/study question
- Impact and relevance for pediatric emergency medicine
- Study hypothesis(es)
- Specific aims(s) (no more than 3 aims)
- Clearly defined and measurable outcome measures - both primary and secondary
- Research approach and methods - include power calculation to justify sample size and analysis plan
- Preliminary data (if applicable)
Add the following sections to the end of your concept proposal (not included in the 3-page limit):
- Narrative budget justification
- References
In addition to your PDF, your application must include:
- Brief abstract (max. 200 words)
- NIH-style biosketches for key personnel (max. 5 pages each)
- Human Subjects Protection Section (if applicable - click here for the suggested template)
- Letters of support, if desired (only required for mentors on fellow applications)
- Detailed line-item budget, with the project period clearly specified (in Word or Excel format)
- Name, email address and phone number of a financial analyst. A financial analyst is the person who can answer questions about your budget and who should receive a copy of the Notice of Award if your project is selected for funding.
Applications will be evaluated based on the NIH scoring system, which considers a project's overall impact as well as the following individual criteria:
- Significance
- Approach
- Innovation
- Investigator
- Sustainability
The system uses a 9-point scale (whole numbers only) for both overall impact scores and individual review criteria scores. The overall impact score is an independent, standalone score determined by the reviewer’s overall assessment of the application based on all strengths and weaknesses; it is not the sum or average of any other scores. This system enables one major strength to outweigh many minor and correctable weaknesses. Reviewers are encouraged to start with an average score of 5 and use the full range of the rating scale. A score of 5 is a good, medium-impact application.
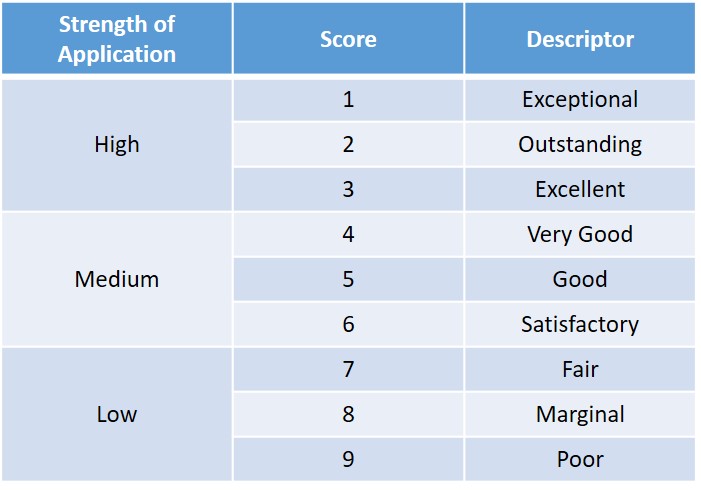
Definitions
- Overall Impact: The project’s likelihood to have an influence on the proposed field of research and, for trainees and junior faculty, on the applicant’s career.
- Significance: Does the research address a problem of high importance or interest in the field?
- Approach: Is the research approach logical and appropriate for the research aims? Are data collection, analysis and interpretation reasonable?
- Innovation: Does the research include a novel idea, method, solution or technology?
- Investigator: Does the investigator’s background suggest s/he will be able to successfully accomplish the stated aims? Will accomplishing this research project enhance the applicant’s career development?
- Sustainability: Sustainability refers to plans for obtaining extramural funding.
Applications are currently being accepted.
All applications must be submitted electronically through the online application portals.
Please make sure you select the correct link: applications cannot be transferred between portals!
General Program & Application Questions
Patricia Bush, M.S., Ed.D.
Senior Research Coordinator
Children’s Healthcare of Atlanta
Emergency Medicine│Egleston Hospital
Office: 404-785-5254
Budget Related Questions
The current financial analyst for the Emory Department of Pediatrics, Division of Emergency Medicine, is Clarrisa Scott.
Frequently Asked Questions
1) For what types of research may these grants be used?
- Funds may be used for any type of pediatric emergency medicine-related research, including basic, clinical, translational, and outcomes research.
- Proposed research must be hypothesis driven and should describe a discrete project that can be accomplished in the time and budget allotted.
- Proposed research must be realistic and achievable within the project period and the requested budget. The most common criticism of proposals from new scientists is over-ambition; your project must be realistic and achievable. You may include a section on future research directions if you wish.
2) I am an adjunct faculty member in Emory's Division of Pediatric Emergency Medicine. Am I eligible to apply for this award?
- No, adjunct faculty members are not eligible for this award.
3) What can I include in my budget?
- You may include whatever is necessary to accomplish your research as long as it is well-justified. Common expenses to include in a research project budget include but are not limited to:
- Personnel (e.g. research coordinator time, data entry assistance)
- Laboratory supplies
- Clinical research costs (e.g. patient engagement fees)
- Core services
- Instrument fees (e.g. fees associated with using a validated scale)
- Publication costs
- It is critical that each budget component be well-justified. A good budget justification clearly explains how the funds will be used in the proposed research project and why they are necessary to complete the project.
4) Can I use a research core located at another institution?
- Yes! While we encourage you to use cores in the Emory Department of Pediatrics or other departments at Emory, you may include cores from other institutions.
5) Does this application require institutional sign-off and do I need to notify RAS of my intent to submit an application?
- Institutional signoff is not required and you do not need to notify RAS of your application, but you do need to work with your division financial analyst on the budget .
6) Do I need an an eRA Commons ID to apply for a GEM$ Award?
- No, you do not need an eRA Commons ID.
7) Are any other funds available to help with publication costs?
- Yes. Emory University's Open Access Publishing Fund provides funds to make it easier for Emory authors to publish in open access (OA) journals and books when no alternative funding is available. The goal is to foster the exploration of new and innovative publishing models across research communities. Please click here for more information.
1) I am new to research and grant writing. Where can I find more information and training resources?
- Check out K-Club, a monthly research career development seminar. Recordings and slides from all previous K-Clubs are available for easy access on the webpage.
- Anyone conducting child health resesearch at Emory is invited to use the Pediatric Cores.
- See this helpful presentation by Emory neonatologist Dr. Ravi Patel on Framing the Research Question - Presentation & Slides
2) What should be included in the abstract?
- Include: the project’s broad, long-term objectives and specific aims, and a description of the research design and method.
- Approach it as a brief summary of what you are planning, why it is important, and if your hypothesis is correct, what you will be able to conclude at the end of the grant and possible next steps.
- Keep in mind that when a grant gets funded, it is usually the abstract that gets posted for public consumption so that people can get a general idea for what you’ve proposed and why it is a good use of funds (i.e. the potential impact of the findings).
- More information is available on this NIH webpage.
3) How do I create a biosketch?
- Biosketch Instructions
- Example Biosketch for Postdoctoral Researchers
- See this helpful presentation on preparing your biosketch by Dr. Stacy Heilman, Director of Pediatric Research Operations
4) Are there examples of narrative budget justifications?
5) Is there someone who can review and provide feedback on my application before I submit it?
- Yes, the Department of Pediatrics Grant Editing and Manuscript Support (GEMS) Core can review and assist with editing your application. Requests received at least 2 weeks before the grant application deadline are guaranteed to be reviewed and edited. You may submit a request less than two weeks before the grant application deadline, but staff may not have time to review your request and the amount of editing will be less than if you submitted it earlier.
1) Do I need IRB approval for my project before submitting my application?
- No, but you will need IRB approval prior to starting any project selected for award.
- No Notices of Award will be sent to any projects involving human subjects research before an IRB approval letter is submitted.
2) How do I know if my project needs to be submitted to the IRB?
- If you are unsure whether your project is human subjects research, we recommend that you request an official determination from the IRB.
- At Children's, complete the Non-Human Subjects Research Determination form and submit it with a brief summary of the project.
- At Emory, request a determination here.
- The following questions may help you determine whether a project needs to be reviewed by the IRB:
- Is the project research - a systematic investigation designed to contribute to generalizable knowledge? The NIH definition of clinical research is available here. If yes or uncertain, submit to the IRB. If no, no submission is required to the IRB.
- Is your project research with human subjects - a living individual about whom you are collecting identifiable information? If yes or uncertain, submit to the IRB. If no, no submission is required to the IRB.
- Potential IRB determinations may include:
- The project does not require IRB review because it does not meet the definition of research with human subjects or clinical investigations.
- The project is human subjects research, but is exempt from further IRB review and approval because it meets the criteria for exemption under 45 CFR 46.101(b). Explanations of exempt categories are available here. An example of a project that may result in this determination is a retrospective chart review using data previously collected for research purposes. Only an IRB can determine if a project meets the criteria for exemption.
- The project qualifies for expedited IRB review. Explanations of expedited categories are available here. Only an IRB can determine if a project meets the criteria for expedited review.
- The project requires full IRB review.
- The HHS Office for Human Research Protections' decision chart provides a helpful visual.
3) My project uses human specimens collected under a protocol for a different study that included a statement in the consent form that gave permission for the specimens to be used for future research. Is my project still considered human subjects research?
- Yes, your project is still considered human subjects research. Answer yes to the human subjects question, and be prepared to provide a copy of the IRB approval letter if your project is selected for funding.
4) Where can I find more information about human subjects research?