Overview
The Integrated Cellular Imaging Core (ICI) provides access to cutting-edge cellular imaging technologies and technical expertise to support pediatric research
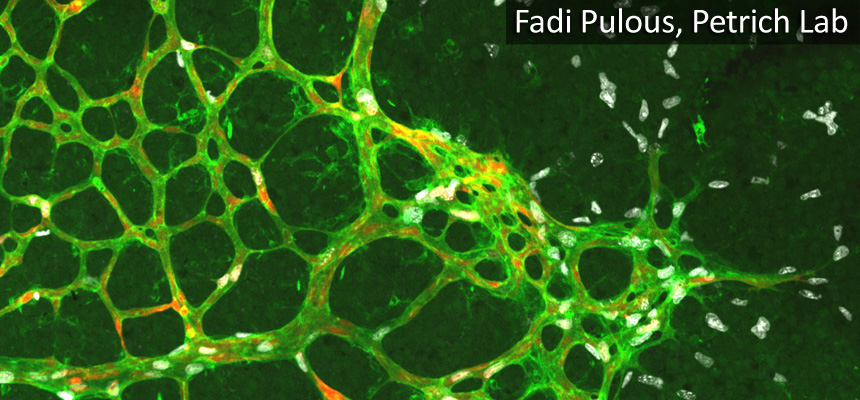
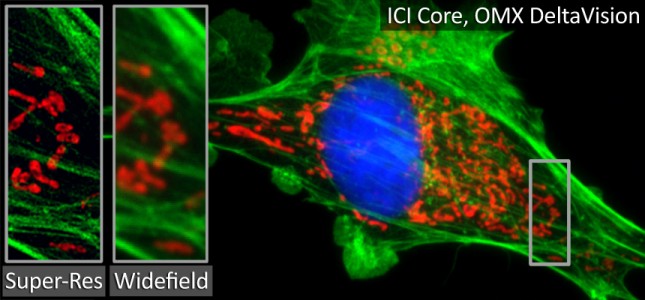
ICI is a partnership facilitated by the Emory School of Medicine and Winship Cancer Institute and includes the Pediatric Research Alliance Cellular Imaging Core. The scientific direction of ICI is led by Scientific Director Dr. Michael Koval, PhD and Core Director Laura Fox-Goharioon. The Health Sciences Research Building I (HSRB I) site is managed by Dr. Hunter Hakimian, PhD and HSRB II is managed by Dr. April Reedy, PhD. Their efforts will help ensure all cellular microscopy research at Emory generate maximum quality data with the highest scientific impact possible. Additionally, ICI organizes educational events and seminars to facilitate scientific exchanges. Please contact them at ici@emory.edu if you would like to be included on the ICI listserv to learn about these events and to receive other information relevant to cellular microscopy.
The pediatric component of the Integrated Cellular Imaging Core is located in the Emory Health Sciences Research Building I (HSRB I) on the ground floor, E-G21 and E310 in HSRB II.
The rates for all the microscopes included in this effort are located on the pricing page. Please click here for details on pricing and subsidy.
Bookings can be made online for the scopes through PPMS. To view the calendars, please click here. Pediatric users are also welcome to use microscopes at any of the ICI locations.
ICI is one of the Emory Integrated Core Facilities (EICF), to learn more about EICF and other core services please see here.
For further assistance regarding this core, please contact ici@emory.edu.
The pediatric component of the Integrated Cellular Imaging Core is located in the Emory Health Sciences Research Building (HSRB) on the ground floor, E-G21.
Bookings can be made online for the scopes through PPMS. To view the calendars, please click here. Pediatric users are also welcome to use microscopes at any of the ICI locations.
For further assistance regarding this core, please contact ici@emory.edu
HSRB I & II Microscopes:
Nikon CSU-W1 SoRa Spinning Disk / HSRB I
The CSU-W1 SoRa is a confocal field scanning spinning disk system and a confocal super resolution system in a single package. We offer dry as well as water immersion lenses for high resolution imaging with fixed and live cell.
- Fast and gentle spinning disk confocal
- two disks, one for confocal, one for super resolution
- easy switch between the two disks
- Four laser lines [nm]: 385, 475, 550, and 621
- Z-stacks, timelapse, multi-point imaging, tiling
- Live-cell imaging with stage top incubator
Leica STELLARIS 8 / HSRB II
- White light laser, extended into the near IR allowing for the application and separation an extended range of spectrally overlapping fluorophores.
- Point scanning inverted confocal
- Galvano and resonant scanners
- 5 detectors: 3 Power HyD-S and 2 Power HyD-X
- Tokai Hit stage top incubator for CO2 and temperature control
- Whitlight lasers, 440-790
- Navigator software for tiling
- TauSense for FLIM
- LIGHTNING for optimal extraction of image details and maximum resolution
Miltenyi UltraMicroscope Blaze Lightsheet / HSRB II
A fully automated light sheet microscope for imaging large or multiple cleared samples at subcellular resolution.
- Super resolution achieved through the clearing of tissue, organs or small animals allowing for complete 3D mapping at single cell resolution.
Leica Thunder Widefield / HSRB II
3D live cell wide field microscope. The Thunder imager helps to maintain optimal physiological conditions by minimizing photobleaching, providing high performance imaging.
- fully motorized DMi8 microscope
- highly sensitive K8 camera
- multi-line, high-intensity fluorescence LED light source
- Live cell imaging with a Tokai Hit stage top incubator
- Computational clearing to minimize out of focus blur
ICI provides state-of-the-art microscopes and support across four satellite locations on campus. For additional details, please click here.
A wide range of microscope are available, allowing researchers an almost endless range of imaging options. Please let us know if you have any questions about what microscopes and techniques would be best for your experiments, email ici@emory.edu
Types of Imaging:
- Confocal
- Live Cell
- Widefield
- Multiphoton
- Super Resolution
- Small Animal Imaging System
- Roche xCELLigence Plate Reader
Please click here to view the ICI rates.
The Integrated Cellular Imaging Core is generously supported by Children's Healthcare of Atlanta and Emory University. When presenting or publishing work completed using the core, please include "Children's Healthcare of Atlanta and Emory University's Pediatric Integrated Cellular Imaging Core" in the acknowledgments.
To determine if you should use “Children’s Healthcare of Atlanta” in your author affiliations, please see guidelines here.
"The Next Level of Fluorescence"
Awardees
Edwin Horwitz: Fluorescence Microscopy Characterization of γMSC phagocytosis
Rabindra Tirouvanziam: Imaging PD1 and Extracellular Vesicle Signaling in Cystic Fibrosis
Curtis Henry: Adipocyte-secreted factors affect chronic inflammation in Acutelymphoblastic leukemia
Crystal Naudin (Rheinallt Jones): 3-dimensional fibrotic scar reconstruction by immunofluorescent microscopy of whole-mounted mouse left ventricular tissue
Brian Petrich: Localization pattern of b1 integrin and talin at endothelial cell-cell junction

