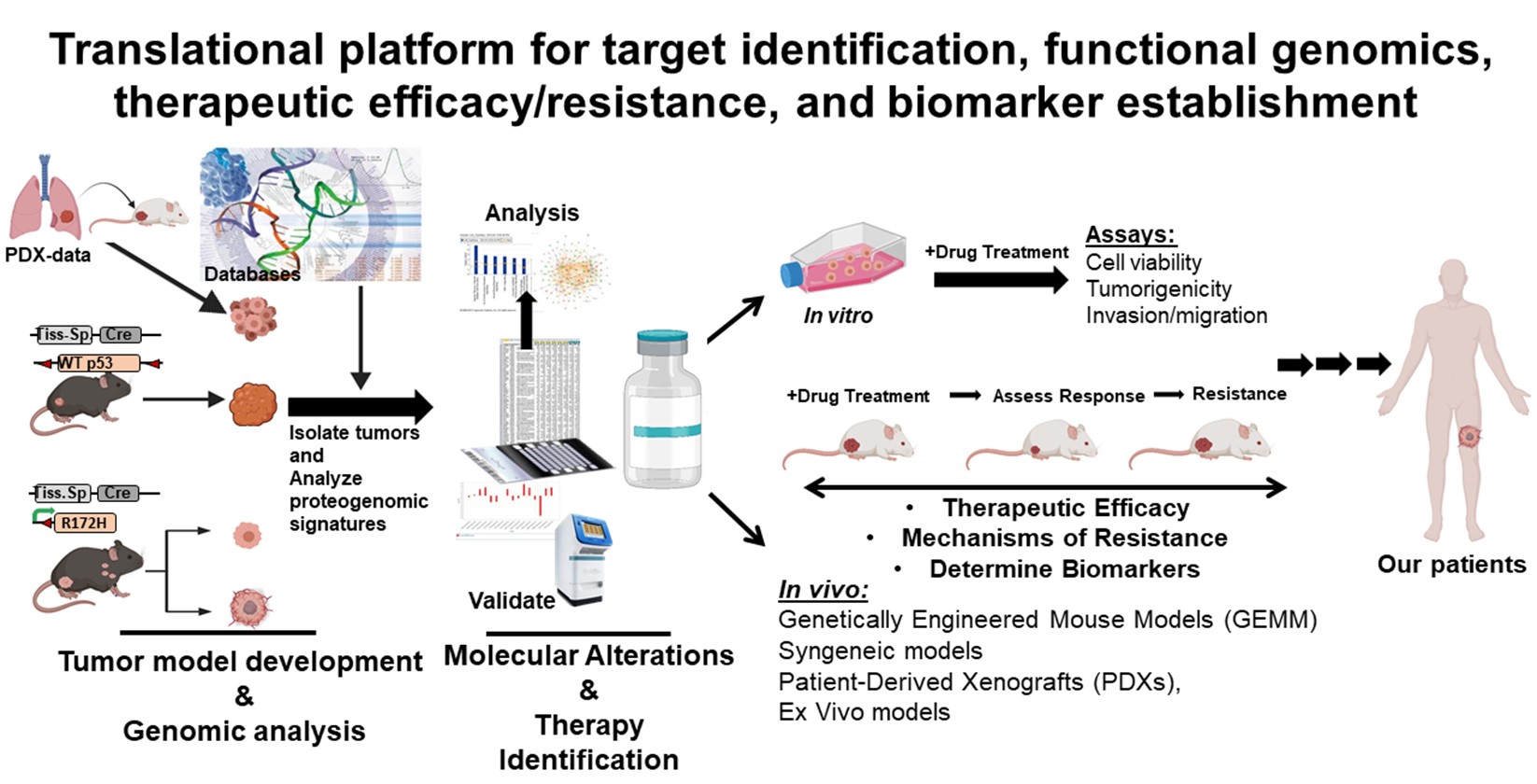
Overview of Research
Due to the relatively poor long-term survival rates, we recognize the need for improving our understanding of the molecular pathogenesis of sarcomas and the identification of new therapeutic avenues. Our translational research lab is at the forefront of studying the pediatric sarcomas, with an emphasis on further understanding the biological, molecular and genetic mechanisms of metastasis and therapeutic resistance for osteosarcoma (OS), Ewing sarcoma (ES), and rhabdomyosarcoma (RMS). There has been minimal progress in improving patient outcomes for these malignancies over the past 20-30 years. My laboratory has tremendous interest and experience in merging innovative murine models, including novel genetically engineered mouse models of OS and RMS, and patient-derived resources towards garnering molecular insights into sarcoma initiation, development/resistance and metastatic progression and translating these findings towards testing novel therapeutic interventions for these aggressive malignancies. For example, our team provided the vital pre-clinical studies essential for developing a clinical trial for Tegavivint, a novel TBL1/β-catenin inhibitor, in the treatment of relapsed pediatric solid tumors, including sarcomas, which opened in October 2021. Our overall goals are to use our extensive models towards gaining critical insights into both tumor intrinsic and extrinsic (e.g., immune and stromal cells) features that will lead to a better understanding of mechanisms of metastasis and therapeutic resistance. We aim to directly translate these insights into identification of pharmacological vulnerabilities that can lead to new treatment options for our patients afflicted with these deadly diseases.

Taku Yamamichi, Post-Doctoral Fellow
Education: MD, PhD (Medicine); Graduate School of Medicine, Osaka University
Current Project: Mechanisms of metastasis in pedatric sarcomas
Fun Fact: Enjoys slow jogging
Email: taku.yamamichi@emory.edu

Bikesh Kumar Nirala, Associate Research Scientist
Education: PhD (Biomedical Engineering), Indian Institute of Technology
Current Project: Development and characterization of the osteosarcoma mouse model
Fun Fact: Not present in the lab and photography
Email: bikesh.kumar.nirala@emory.edu

Tajhal D. Patel, Staff Scientist
Education: PhD (Cell and Molecular Biology), Baylor College of Medicine
Current Project: Utilizing bioinformatic approaches to study pediatric sarcomas
Fun Fact: Mom to triplets
Email: dayaram@bcm.edu

Juan Dou, Senior Research Specialist
Education: Masters (Philosophy in Pediatrics), Sun Yat-sen University of the third affiliated hospital, Guangzhou, China, 2006; MD (Medical Sciences), Wannan Medical College, Wuhu, Anhui, China, 1994
Current Project: Molecular Mechanisms underlying Rhabdomyosarcoma (RMS) and Osteosarcoma (OS) and Mechanism-based therapeutics for these children-associated solid tumors
Fun Fact: Enjoy reading, cooking, gardening and traveling
Email: jdou2@emory.edu

Tasnuva Nuhat Shafin, Research Specialist
Education: MS (Physiology and Neurobiology), University of Connecticut
Current Project: Use of combinatorial therapies to improve immune-mediated approaches for high-risk osteosarcoma
Fun Fact: Enjoys cooking, traveling and spending time with friends and family
Email: tshafin@emory.edu

Sarah S. Kappa, Pedaitric Hematology/Oncology Fellow
Education: MD, Emory University School of Medicine; Residency and Chief Residency, Children's National Hospital, Washington DC; Fellowship, Texas Children's Hospital/Baylor College of Medicine, Houston, TX
Current Project: Investigating Oncogenic Signaling Inhibition in Combination with Immune Checkpoint Blockade for the Treatment of Osteosarcoma
Fun Fact: When I studied abroad during college I bungy jumped off of the Bloukrans Bridge in South Africa, which is one of the world's highest bungy jumps. While it was exhilarating, it is likely not something I will do again!
Email: sskappa@texaschildrens.org
Tsang, S.V., N. Rainusso, M. Liu, M. Nomura, T. D. Patel, K. Nakahata, H.R. Kim, S. Huang, K. Rajapakshe, C. Coarfa, T.-K. Man, P. H. Rao, and J. T. Yustein. “LncRNA PVT-1 promotes osteosarcoma cancer stem-like properties through interaction with TRIM28 and TSC2 ubiquitination”. Oncogene. 2022 Nov 8. doi: 10.1038/s41388-022-02538-w. Online ahead of print. PMID: 36348010
Tu, J., Z. Huo, Y. Yu, H. Luo, D. Zhu, A. Xu, R. Wang, R. Hu, J.A. Gingold, J. Su, M.F. Huang, K.L. Tsai, R. Zhou, H.M. Chen, W. He, S.H. Chen, T.R. Webb, H. Yang, P. Jia, J.T. Yustein, L.L. Wang, M.C. Hung, Z. Zhao, R. Zhao, C. D. Huff, J. Shen, and D.F. Lee. “Hereditary retinoblastoma iPSC mode reveals spliceosome as a therapeutic vulnerability of RB1-mutant cancers”. Proc Natl Acad Sci U S A. 2022 Apr 19;119(16):e2117857119. doi: 10.1073/pnas.2117857119. Epub 2022 Apr 11.PMID: 3541290
Guo, H. Huang, Z. Zhu, M. J. Chen, H. Shi, P. Sharma, J. P. Connelly, S. Liedmann, Y. Dhungana, Z. Li, D. Haydar, M. Yang, H. Beere, J. T. Yustein, C. DeRenzo, S. M. Pruett-Miller, G. Krenciute, C. W.M. Roberts, H. Chi, D. R. Green. “The SWI/SNF canonical BAF complex and c-Myc cooperate to promote early fate decisions in CD8+ T cells” Nature. 2022 Jul;607(7917):135-141. doi: 10.1038/s41586-022-04849-0. Epub 2022 Jun 22. PMID: 35732731
K. Nakahata, B. Simons, E. Pozzo, R. Shuck, L. Kurenbekova, T. Patel, and J. T. Yustein. “MyoD-Cre driven alterations in mutant Kras and p53 leads to a mouse model with histological and molecular characteristics of human rhabdomyosarcoma with pre-clinical applications” Dis Model Mech. 2022 Feb 1;15(2):dmm049004. doi: 10.1242/dmm.049004. Epub 2022 Feb 17.PMID: 35174853 Editor’s Choice; Highlighted in special Ras Issue due out in 02/2022.
Liikanen, I., C. Lauhan, S. Quon, K. Omulisik, A.T. Phan, L. Barcelo- Bartroli, J. Goulding, J. Chen, J. P. Scott-Browne , J.T Yustein, N. E. Scharping, D.A. Witherden, A.W. Goldrath. “Hypoxia-inducible factor activity promotes anti-tumor effector function and tissue-residency by CD8+ T cells”. J Clin Invest. 2021 Apr 1;131(7):e143729. doi: 10.1172/JCI143729. PMID: 33792560
Shawki L. Qasim, Laura Sierra, Ryan Shuck, Lyazat Kurenbekova, Tajhal D. Patel, Kimal Rajapakshe, Jade Wulff, Kengo Nakahata, Ha Ram Kim, Yosef Landesman, TJ Unger, Cristian Coarfa, and Jason T. Yustein. “p21-Activated Kinases as a viable therapeutic target for high-risk Ewing sarcoma”. Oncogene. 2021 Jan 7. doi: 10.1038/s41388-020-01600-9. PMID: 33414491
Dasgupta, A., L. Sierra, S. Tseng, L. Kurenbekova, T. Patel, R. Shuck, N. Rainusso, C. Coarfa and J. T. Yustein. “Targeting PAK4 has therapeutic potential for relapsed and metastatic rhabdomyosarcoma”. Cancer Res. 2020 Nov 9:canres.0854.2020. doi: 10.1158/0008-5472.CAN-20-0854. Online ahead of print. PMID: 33168646
Motonari Nomura, Nino Rainusso, Yi-Chien Lee, Brian Dawson, Ruolan Han, Jeffrey L. Larson, Ryan Shuck, Lyazat Kurenbekova and J.T. Yustein. “Tegavivint and the β-catenin/ALDH Axis in Chemotherapy-Resistant and Metastatic Osteosarcoma”. J Natl Cancer Inst. 2019 Feb 21. pii: djz026. doi: 10.1093/jnci/djz026.
NIH/R01
Dissecting and Targeting Oncogenic Functions of PAK4 in High-Risk Rhabdomyosarcoma
Role: PI
NIH/R01
Intratumoral Imaging of Hypoxia Using 1H- and 19F-MRI with Redox-Responsive Eu-Based Contrast Agents
Role: PI
MPI: Allen/Pautler
The Charlie Landers Foundation
Dissecting and Targeting PAK4-Mediated Signaling in Ewing Sarcoma Development and Metastasis
Role: PI
NIH/R21
Development of Bone-Targeting Antibodies for Ewing Sarcoma Using Genetic Code Expansion
Role: PI
MPI: Xiao
NIH/R21
Dissecting and Targeting the Role of GALNT14 in High-Risk Osteosarcoma Development and Progression
Role: PI
MPI: Kohler
Innovations in Oncology Award
Characterizing and Targeting Intrinsic and Immune Modulatory Functions of PAK4 in High-Risk Rhabdomyosarcoma
Role: PI
Making It Better (MIB) Research Award
Combinatorial Therapies to Improve Immune-Mediated Approaches for Osteosarcoma
Role: PI
Osteosarcoma Institute
Use of Combinatorial Therapies to Improve Immune-Mediated Approaches for High-Risk Osteosarcoma
Role: PI
MPI: Gottschalk
Rally Foundation for Childhood Cancer Research
Dissecting and Targeting the WNT Pathway in Metastatic Osteosarcoma
Role: PI