This Core provides consultation assistance and can run queries, compile data, and conduct analyses for investigators wishing to perform outcome studies related to pediatric heart diseases. All requests will be subject to review/approval to ensure match with an outcomes research scope and to ensure all compliance requirements are met.
Collection of these data was made possible by the Pediatric Cardiac Care Consortium (PCCC), a non-profit foundation whose mission is to improve the lives of patients with pediatric acquired and congenital heart disease by answering key questions about diagnoses, treatments, and outcomes, and thereby empower patients, families, clinicians, and policy-makers to make informed decisions and improve quality of care. Dr. Lazaros Kochilas, President of PCCC, will review all data requests submitted to the core and will provide approval contingent upon meeting specified criteria, including confirmation of the proper scope of data use (i.e. outcomes research) and adherence to all compliance-related requirements.
Learn more about the PCCC here.
The rates are calculated based on the cost of the operations and maintenance of the core. The prices listed for the base fee and analysis rate are for internal investigators. External collaborators will need to contact the core for a quote. A quote will be provided at the initiation of a new project. The final invoice will be issued upon completion of the project. For an investigator requesting an additional project using the same cohort, only the analysis rate will be charged. A new base fee will be charged for any request requiring a new cohort.
- The Base fee is $3,377
- Analysis rate is $75/hour
Please contact the PHDD Registry Core to discuss using the PCCC database. Full detail, including a complete list of existing variables for analysis, is available upon request.
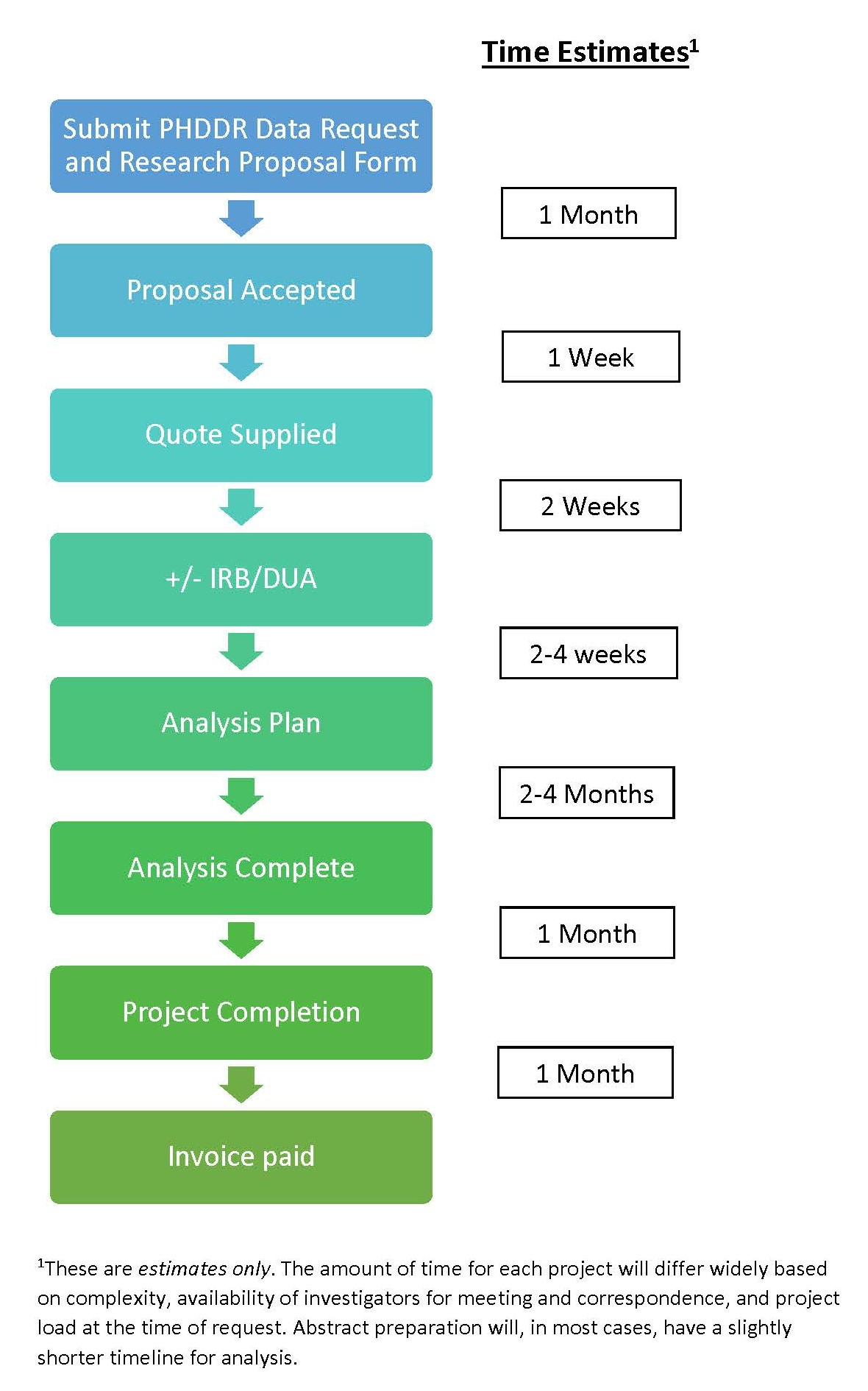
When ready, complete the PHDDR Data Request and Research Proposal Form to use our services for your project. Once your project has been approved, you may be required to obtain approval from the Institutional Review Board (IRB). For external users, depending on the level of access required to conduct your research, there may be additional requirements. The Core will assist you with these requirements.
Please see the timeline of how our service works with collaborators requesting data. Contact Krisy Kuo for full details including a complete list of variables available for analysis.
The Pediatric Heart Diseases Data Registry Core is graciously supported by Children’s Healthcare of Atlanta and Emory University in collaboration with Participating PCCC Centers. When presenting or publishing work completed using data from the PCCC, please include “Children’s Healthcare of Atlanta and Emory University’s Pediatric Heart Disease Data Registry Core” and the PCCC centers in the acknowledgments. Suggested text:
This study was supported by the efforts of the Children’s Healthcare of Atlanta and Emory University’s Pediatric Heart Disease Data Registry Core. Additionally, we thank the program directors and data collection coordinators from the participating PCCC centers; without their effort and dedication, this work could not have been completed.
If your publication utilizes NDI and/or UNOS linked data then you will need to include additional text, with which the PHDDR Core will assist.



